INCI lists are the standardized ingredient disclosures on every cosmetic product. Here's what the naming system means, how ordering works, and what the list can't tell you.
By Skintrig Team
Skintrig Team publishes practical, privacy-first guides about ingredient tracking and skincare reaction logging.
Most skincare labels look like a wall of Latin. Aqua. Glycerin. Butylene Glycol. Phenoxyethanol. If you’ve ever squinted at the back of a moisturizer and wondered what any of it means, you’ve been looking at an INCI list.
Understanding what that list is — and how to read it — won’t tell you how your skin will respond to a product. But it gives you the vocabulary to start paying attention.
What is an INCI list?
INCI stands for International Nomenclature of Cosmetic Ingredients. It’s a standardized naming system for cosmetic and personal care ingredients, maintained by the Personal Care Products Council (PCPC) and adopted by regulators in the EU, US, and many other markets.
Every cosmetic product sold in most regulated markets is required by law to display its ingredients using INCI names. The list must appear on the packaging — and in the EU, must also be accessible online. The goal of standardization is transparency: the same ingredient should be identifiable across every product, regardless of brand, country, or marketing language.
If you see “Aqua” on a label, that’s INCI for water. “Tocopherol” is vitamin E. “Parfum” is fragrance. The names are consistent across the entire system — which is exactly the point. Once you understand the naming conventions, you can compare ingredient lists across products in a meaningful way.
Why do INCI names look so unfamiliar?
The naming system was designed for technical accuracy, not readability. Most names derive from Latin binomials (for plant extracts), IUPAC chemistry nomenclature (for synthesized compounds), or modified common names. This is why “Cocos Nucifera Oil” appears instead of “coconut oil” — the INCI name specifies the botanical source with scientific precision.
Some ingredients map easily to common language equivalents: Retinol is Retinol, Niacinamide is Niacinamide. Others are less intuitive. “Cetearyl Alcohol” is a fatty alcohol used as an emollient — not the drying kind of alcohol. “Sodium Lauryl Sulfate” is a surfactant, commonly abbreviated SLS.
The unfamiliarity isn’t there to obscure anything. It’s the result of building a global technical standard. Once you recognize a handful of key names, the list starts to become readable. And because the names are consistent, an ingredient you learn to recognize in one product will appear identically in every other product that contains it.
How does ingredient order work?
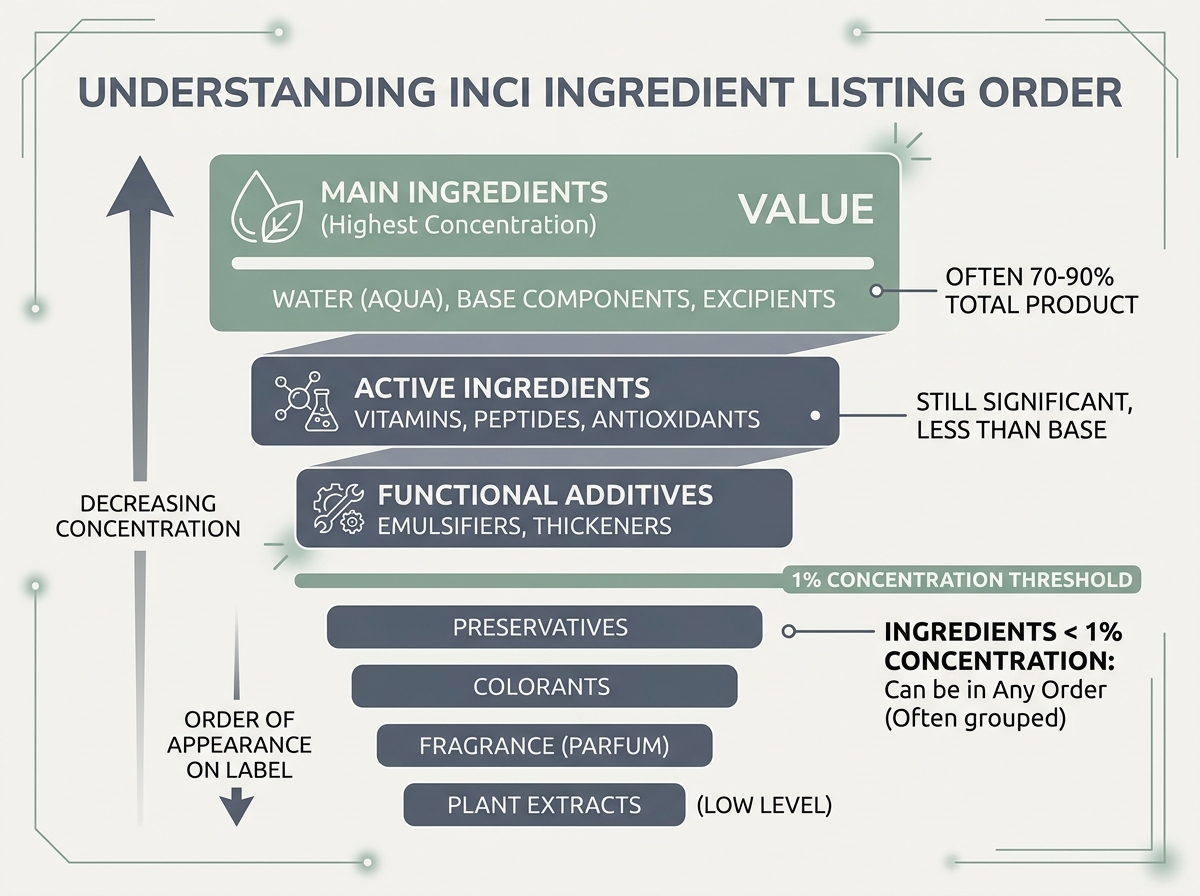
INCI lists follow a specific ordering rule: ingredients are listed in descending order of concentration by weight, from highest to lowest. The ingredient present in the greatest amount appears first. This applies to all ingredients present at concentrations above 1%.
Ingredients present at 1% or below can appear in any order after the above-1% ingredients. This threshold matters. Preservatives, colorants, fragrances, and actives added at low concentrations — such as retinol or certain peptides — may appear at the end of a list without that signaling they’re ineffective. What constitutes an effective concentration varies widely by ingredient.
One practical implication: if “Aqua” is the first ingredient, the formulation is predominantly water-based. If a fatty oil appears first, the product is oil-heavy.
What doesn’t an INCI list tell you?
The INCI list tells you which ingredients are present and roughly in what relative order. It does not tell you exact concentrations — except where regulations require specific disclosure, such as certain actives in medicated products. It doesn’t tell you the source or quality of an ingredient, its supplier, or how it was processed.
It also doesn’t tell you how your skin will respond. Two products with similar INCI lists may perform differently due to pH, formulation interactions, or particle size. Whether you personally react to an ingredient is something no label can predict in advance.
One more nuance worth knowing: “fragrance-free” as a marketing claim is not the same as a product that contains no “Parfum” in its INCI list. Some masking agents may not be disclosed as fragrance. “Parfum” in an INCI list is a regulated disclosure of a fragrance mixture — but individual components within that mixture are typically not itemized on the label.
How does this connect to tracking your own patterns?
Reading INCI lists is the foundation of ingredient tracking. Before you can identify which ingredients appear consistently in products that preceded a reaction, you need to be able to recognize them by name across different products. The INCI system makes that possible — because “Phenoxyethanol” in a toner and “Phenoxyethanol” in a serum are the same thing, regardless of how those products are marketed.
If you’re logging products in Skintrigue, this is where INCI becomes practically useful. When you paste or scan an ingredient list, Skintrigue reads it against your logged reaction history to surface any ingredients that appear frequently in products you’ve reacted to. The pattern — if one exists — shows up in your own data, not in a generalized claim about what’s harmful for reactive skin.
Your data is specific to you. The INCI list is how that specificity becomes searchable.
The practical takeaway
You don’t need to memorize ingredient chemistry to benefit from reading INCI lists. What matters is building enough familiarity to recognize key names across products, and understanding what the list can and can’t tell you. The INCI system gives you consistent vocabulary. What you do with that vocabulary — which ingredients you track, which patterns you notice — is something only your own logged data can answer over time.
FAQ
Is INCI the same in every country?
INCI is used across the EU, US, Canada, Japan, and many other markets, but not universally. In the EU, INCI labeling is mandatory under the Cosmetics Regulation (EC 1223/2009). In the US, the FDA requires cosmetics to list ingredients using INCI names. The system is internationally harmonized for most common ingredients, but some local markets have partial divergences. When comparing products across regions, the INCI list is still the most reliable cross-reference available.
Why does the same ingredient sometimes appear under different names on different products?
This can happen with older products, non-compliant labeling, or ingredients where the INCI name has been updated over time. It also happens when a brand uses a trade name in marketing copy while the regulated label is INCI-compliant. When in doubt, the back-of-pack label is the authoritative disclosure — not ingredient spotlights, packaging fronts, or brand websites.
Can I use an INCI list to know if a product is right for my skin?
An INCI list tells you what’s in a product — not whether it will work for your skin. Whether any ingredient correlates with reactions you’ve experienced is something your own logged history can help clarify over time. An INCI list is a starting point, not a verdict, and individual responses to ingredients vary too much to draw universal conclusions from a label alone.
What’s the difference between an INCI list and a “key ingredients” section?
Key ingredient sections are marketing content, not regulated disclosures. Brands choose which ingredients to spotlight based on what they want to feature — an ingredient highlighted on the front of a box may appear near the bottom of the INCI list in a very small concentration. The INCI list on the back or side of the product is the complete, regulated disclosure of everything in the formulation.
Want these insights in one place?
Join the waitlist to get early access when beta opens (planned for mid to end of March 2026).
Join waitlistRelated posts
-
 Common irritants in skincare: how to identify and avoid your triggers
Common irritants in skincare: how to identify and avoid your triggers A practical guide to the skincare ingredients that most often irritate sensitive skin, how to read labels more carefully, and how to spot patterns in your own routine without guessing.
-
 How to Find What's Causing Your Breakouts (Without Guessing): A Simple Tracking Method
How to Find What's Causing Your Breakouts (Without Guessing): A Simple Tracking Method A practical method to identify your personal skincare triggers using timing + ingredient overlap,plus how INCI scanning and tracking makes it effortless.
-
 Why "Fragrance-Free" Doesn't Mean Irritation-Free
Why "Fragrance-Free" Doesn't Mean Irritation-Free "Fragrance-free" has a specific regulatory meaning that doesn't cover every scent-related ingredient. Here's what the label actually tells you, and what it doesn't.